What is the Best Coagulant Chemical for Water Treatment?
Water treatment is essential for public health and environmental safety. Choosing the right coagulant chemical is a crucial step in this process. Coagulant chemicals help to remove suspended particles in water. They work by neutralizing the charges on particles, allowing them to clump together and settle.
Various coagulant chemicals exist, each with unique properties. Common options include alum, ferric chloride, and polymeric coagulants. These substances differ in effectiveness, cost, and application methods. Understanding their characteristics is key to selecting the most suitable one for specific water conditions.
However, the choice of coagulant chemical is not straightforward. It requires careful consideration of factors like water quality, treatment goals, and operational constraints. Some coagulants may lead to unwanted by-products. Moreover, excessive use can harm aquatic ecosystems. Thus, ongoing research and adaptation to local conditions are vital for effective water treatment.

Definition and Importance of Coagulants in Water Treatment
Coagulants play a crucial role in water treatment. They aid in the removal of suspended particles and improve water clarity. By promoting the aggregation of small particles, coagulants lead to larger clumps called flocs. These flocs are easier to remove during filtration processes. Without effective coagulants, water quality can suffer. This can lead to issues in drinking water safety and environmental impact.
Choosing the right coagulant is key. There are various types, including inorganic and organic coagulants. Each has unique properties and applications. It's important to consider factors like pH, temperature, and the type of contaminants in the water. Sometimes, a combination of chemicals may be necessary. This can complicate the treatment process, requiring careful adjustments and monitoring.
**Tip:** Always perform jar tests before full-scale application. This will help determine the optimal dosage of coagulants needed.
Occasionally, coagulant use can lead to challenges. Overdosing can cause excessive sludge. Conversely, too little may not effectively reduce turbidity. Operators must remain vigilant and adjust as needed. Continuous learning and reviewing past treatments can improve future results.
**Tip:** Maintain detailed records of chemical use and water quality. This data can guide adjustments over time.
Types of Coagulant Chemicals Used in Water Treatment
Coagulation is a vital process in water treatment. It removes suspended particles to improve water clarity. Various coagulant chemicals are employed in this process, each with unique properties and uses.
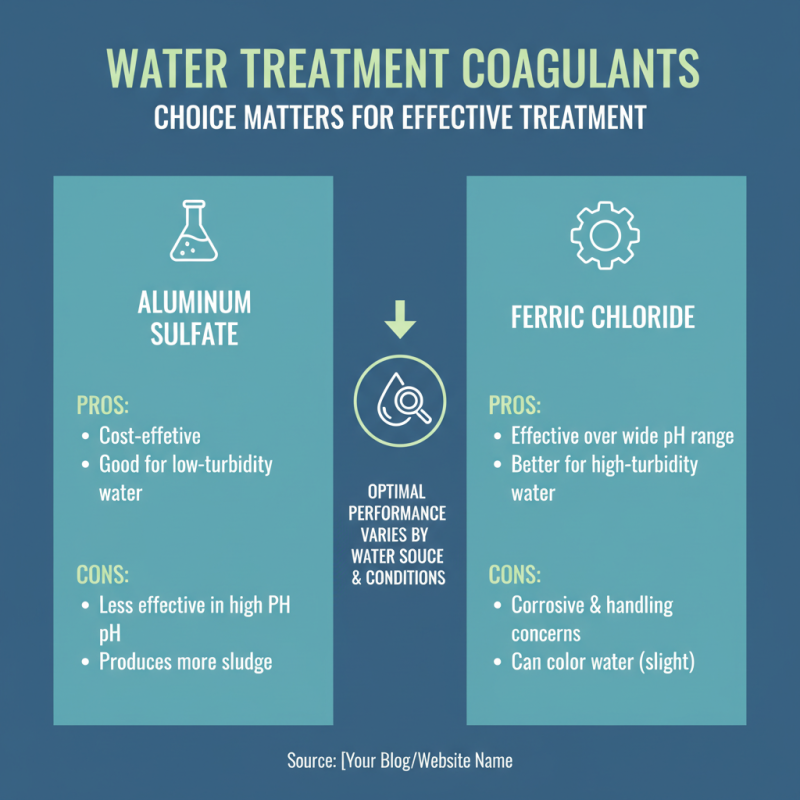
One common type is alum, or aluminum sulfate. It works by neutralizing particle charges, allowing them to clump together. This clumping makes it easier to remove contaminants. Another widely used coagulant is ferric chloride. It is effective in removing organic matter, color, and turbidity. Its mechanism is slightly different, relying on iron ions to bind with particles.
Polymers are also significant in water treatment. They enhance coagulation by providing additional binding sites. This can lead to a stronger floc that is easier to separate. Some facilities are exploring more environmentally friendly options as well. While these alternatives show promise, they often require more testing and adjustment. Finding the right coagulant can be tricky; factors like water quality and treatment goals greatly influence the choice.
What is the Best Coagulant Chemical for Water Treatment? - Types of Coagulant Chemicals Used in Water Treatment
| Coagulant Type |
Chemical Formula |
Common Use |
Advantages |
Disadvantages |
| Aluminum Sulfate |
Al2(SO4)3 |
Drinking water treatment |
Effective for turbidity removal |
May increase aluminum levels in water |
| Ferric Chloride |
FeCl3 |
Wastewater treatment |
High efficiency in floc formation |
Can be corrosive and irritates skin |
| Polyaluminum Chloride |
PAC |
Municipal water treatment |
Less sludge production |
Cost may be higher than alternatives |
| Calcium Hydroxide |
Ca(OH)2 |
Softening of water |
Widely available and low cost |
Larger volumes needed |
| Sodium Aluminate |
NaAlO2 |
Flocculation aid |
Enhances particle removal |
Can affect pH levels significantly |
Factors Influencing the Choice of Coagulant Chemicals
When selecting coagulant chemicals for water treatment, various factors come into play. Water quality is a primary consideration. Different sources of water contain unique impurities. These can affect the choice of coagulants used in the treatment process. For instance, turbid water may require a stronger coagulant to effectively reduce particle load.
Operational conditions are crucial as well. Temperature, pH, and mixing intensity influence coagulation efficiency. Some coagulants work better under specific pH levels. This can significantly impact treatment results. Additionally, dosage levels must be adjusted based on the chemical being used and the characteristics of the water. Finding the right balance can be challenging. Too much coagulant may lead to excessive sludge and operational inefficiencies.
Cost is another important factor that cannot be overlooked. Budget constraints often dictate the choice of coagulants. However, opting for cheaper alternatives might compromise water quality. This trade-off requires careful consideration. Environmental effects of the chosen coagulant also matter. The goal is to achieve effective treatment while minimizing negative impacts. Balancing all these factors is essential for successful water treatment processes.
Comparative Effectiveness of Common Coagulants
When it comes to water treatment, the choice of coagulant chemicals can greatly impact effectiveness. Different coagulants may perform well under various conditions. Common options include aluminum sulfate and ferric chloride. Each has its strengths and weaknesses in different water sources.
Aluminum sulfate is often favored for its efficiency. It binds well with colloidal particles, helping to reduce turbidity. However, its effectiveness can diminish in extremely alkaline conditions. Ferric chloride, on the other hand, works efficiently in a wider range of pH levels. Its performance, though good, can be hindered by high organic loads in the water.
Other coagulants, like polyaluminum chloride, are gaining attention. They can provide rapid settling and fewer residuals. Still, they may not be universally applicable. Knowing when and where to apply different coagulants requires careful consideration and field testing. Not every coagulant will suit every situation. Factors like cost, availability, and local water chemistry play significant roles. Making the right choice demands awareness of these complexities.
Environmental Considerations and Regulations in Coagulation Processes
Coagulation is a critical process in water treatment. It involves adding chemicals to remove impurities. Environmental impacts cannot be overlooked. Many coagulants can release harmful residues. These substances may affect aquatic life. Regulators often set strict limits on these chemicals.
Choosing the right coagulant requires balancing efficacy and environmental safety. Aluminum sulfate and ferric chloride are commonly used. However, they can have negative effects on ecosystems. Proper disposal of sludge is crucial. Waste management practices must align with environmental standards.
Communities face challenges in monitoring these processes. Not every facility has the resources needed for compliance. Regular training for staff is essential. This ensures adherence to regulations. Yet, many still struggle with this aspect. Continuous improvement is vital for sustainable water treatment.
Best Coagulant Chemicals for Water Treatment
This bar chart illustrates the effectiveness of various coagulant chemicals used in water treatment processes, measured in terms of their removal rates. Aluminum Sulfate shows the highest effectiveness in removing impurities, followed by Ferric Chloride and Polymeric Coagulants.